For decades, industrial quality control has been locked in a rigid cycle: pull a sample, send it to the lab, wait for the results, and hope the batch hasn’t drifted out of spec in the meantime.
Today, that model is obsolete.
Driven by the demand for Continuous Manufacturing and Quality by Design (QbD), process analytics are moving out of the laboratory and directly into the manufacturing line. At the forefront of this shift is VIAVI’s MicroNIR spectrometer family.
By delivering non-destructive, molecular-level analysis in milliseconds, MicroNIR instruments are redefining what it means to control a process. While the technology is utilized across a massive range of industries, its impact is perhaps most revolutionary in pharmaceutical manufacturing.
Here is a look at how VIAVI’s MicroNIR portfolio is being deployed across the modern manufacturing landscape.
Beyond Pharma: A Cross-Industry Workhorse
Before diving into the pharmaceutical applications, it is worth noting the sheer versatility of the MicroNIR platform. Because near-infrared (NIR) spectroscopy provides fast, non-contact analysis of organic compounds with zero sample preparation, these instruments ranging from the rugged OnSite handhelds to process-integrated PAT units are highly adaptable.
- Food, Feed & Agriculture: Used both in the field and online to instantly quantify moisture, protein, fat, and sugar content in everything from raw sugarcane to processed cheese.
- Chemical & Petrochemical: Deployed inline for reaction monitoring, solvent composition tracking, and polymer production control.
- Field Security & Law Enforcement: Utilized as portable, wireless units by border patrol and law enforcement for the rapid, non-invasive identification of narcotics and explosives.
- General Industrial Control: Integrated directly into equipment loops to reduce cycle times and eliminate blind spots in standard manufacturing.
Revolutionizing Pharmaceutical Manufacturing
While VIAVI serves multiple sectors, the MicroNIR platform is purpose-built to conquer the complexities of pharmaceutical manufacturing. By enabling Process Analytical Technology (PAT), MicroNIR allows drug manufacturers to replace slow offline assays (like HPLC or LOD) with real-time process control.
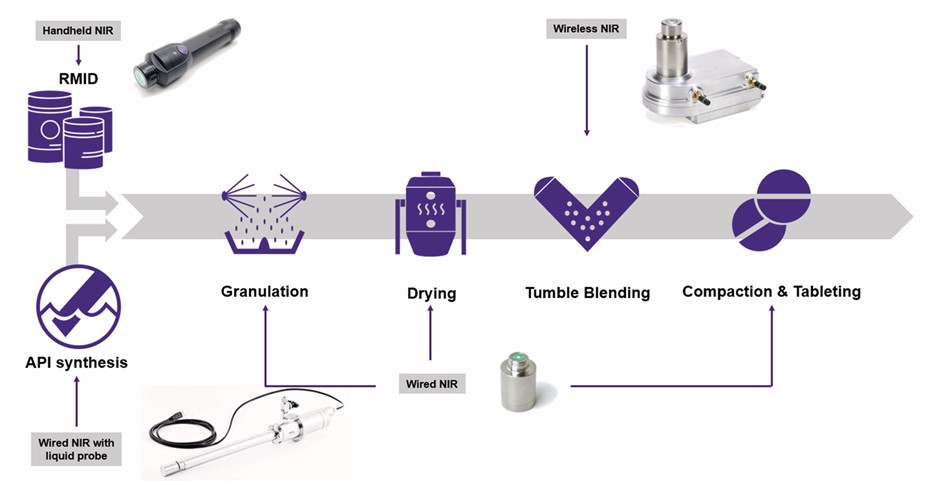
Here is how MicroNIR integrates into the critical unit operations of a modern solid-dose and liquid API pharmaceutical line.
- Pre-Manufacturing: Raw Material ID (RMID)
Quality starts before the process even begins. Using handheld units or at-line sampling stations, operators can perform instant spectral fingerprinting to verify APIs, differentiate excipients (like lactose or starch variants), and flag counterfeit materials. It can even provide proxy measurements for moisture content and bulk density before materials enter the blender.
- Midstream Processing: The Core of Solid Dose
The most high-value use cases for MicroNIR exist in the transformation of raw powders into ready-to-press granules.

- Blending: Integrated into tumble or bin blenders, MicroNIR provides real-time tracking of Blend Uniformity (BU) and Content Uniformity (CU). By using Moving Block Standard Deviation (MBSD) algorithms, the system can pinpoint the exact moment of blend homogeneity, preventing segregation and optimizing the cycle time.
- Granulation: Whether monitoring wet granulation (moisture content, binder distribution) or dry roller compaction (ribbon density, API concentration), NIR enables closed-loop control of the equipment, providing immediate feedback to spray rates or impeller speeds.
- Drying: Water has a distinct O-H absorbance in the near-infrared spectrum. By continuously monitoring the drying curve in a fluid bed, MicroNIR serves as a real-time replacement for traditional Loss-on-Drying (LOD) tests. This actively prevents the batch-ruining risks of over-drying (tablet hardness issues) or under-drying (stability risks).
- Downstream Processing: Compression to Coating
As the product reaches its final physical form, MicroNIR continues to monitor Critical Quality Attributes (CQAs). By monitoring the feed frame, it ensures the blend uniformity feeding the press is consistent. During the coating process, it tracks layer growth, coating uniformity, and residual moisture, pinpointing the precise endpoint of the coating cycle.
The Engine Behind the Hardware: Chemometrics & Compliance
Hardware alone does not secure regulatory approval. The true engine of the VIAVI MicroNIR portfolio in a GMP environment is its data and modelling infrastructure.
The platform supports a seamless transition from R&D to full production. Models built in the lab using Partial Least Squares (PLS) for quantification or Principal Component Analysis (PCA) for anomaly detection can be directly transferred to pilot and production lines.
Crucially, the software layer is built for strict regulatory environments, offering full 21 CFR Part 11 compliance, robust audit trails, and instrument qualification protocols (USP <1119>, EP 2.2.40).

The Bottom Line
VIAVI’s MicroNIR portfolio is not just about taking measurements; it is about fundamentally changing the manufacturing workflow.
By integrating composition analysis and material identification directly into the process line, manufacturers can unlock Real-Time Release Testing (RTRT). The result? Drastically reduced cycle times minimized batch failures, and a seamless path to the future of continuous manufacturing.
List of what is possible with our NIR sensors:
✅ Raw materials | ✅ Blending |
API and excipient identification | Blend uniformity |
✅ Granulation | ✅ Drying |
Moisture during granulation | Residual moisture |
✅ Milling / particle size | ✅ Compression |
Particle size estimation | API content per tablet |
✅ Coating | ✅ API synthesis (small molecules) |
Coating thickness | Reaction monitoring |
✅ Crystallisation | ✅ Final product |
Concentration monitoring | Content uniformity verification |
To learn more, check out the MicroNIR Product Range or contact our team at info@antech.ie.
